Emicizumab- The game changer in Haemophilia
- ACS BCP
- Feb 22
- 3 min read
Haemophilia, ‘the royal disease’, is a rare & serious disorder affecting thousands of people around the world, running from centuries, right from the Queen Victoria of England to an ordinary boy living across the street.
Many people with Haemophilia are undiagnosed or poorly treated.
Haemophilia is an X-linked recessive disorder, with the X-chromosome carrying defective/mutated genes encoding for clotting factors. Haemophilia A and B manifest due to deficiency of factor VIII & factor IX respectively. Haemophilia C, which is rare, is due to deficiency of factor XI.
Like all other X-linked diseases, if only one of the X-chromosomes carries the mutation, the female is said to be a carrier for the disease, and if the X-chromosome in a male carries the mutation, the male is said to be diseased.
The clotting pathway in humans is broadly divided into: intrinsic pathway and the extrinsic pathway. Both pathways consist of a series of cascade enzyme activation events that lead to the formation and stabilization of a blood clot by crosslinking of fibrin monomers and activation of platelets. Both pathways are explained in the adjoining figure.
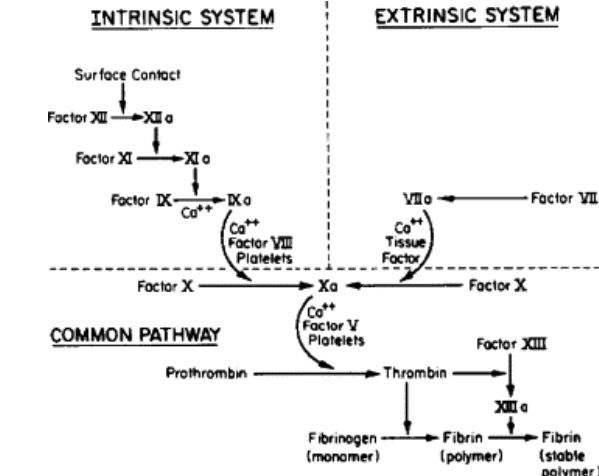
Hemophilia usually presents as bleeding after minor trauma or as a spontaneous bleed. Any patient with hemophilia who presents with a severe acute bleeding episode requires quick recognition of the location and severity of the bleed; this must be followed by immediate replacement with high-dose clotting factor concentrate (CFC) with factor VIII or IX.
An important consideration in the treatment for haemophilia is the formation of inhibitors against the clotting factor infusions. Inhibitors are neutralizing antibodies produced by the patient upon infusion of clotting factors. These antibodies are usually IgG antibodies, which bind to and neutralise the activity of clotting factors, rendering the standard treatment ineffective, making it difficult to control bleeding.

Alternative treatments include:
1. Immune Tolerance Induction (ITI): In this method, frequent and regular exposure to factor VIII concentrate is made, as a way to induce tolerance towards the same. This occurs through T-cell exhaustion due to overstimulation, leading to T-cell anergy, where T-cells become unresponsive to exposed antigens.
2. FEIBA: FEIBA stands for Factor VIII Bypassing Agent, which is FEIBA is an activated prothrombin complex concentrate (aPCC)containing activated Factor VII (FVIIa) and non-activated Factors II, IX, and X. These components bypass the need for Factor VIII to form a clot, which is crucial when inhibitors have neutralized Factor VIII.
The current progress in the treatment methods is the use of Emicizumab for the treatment of haemophilia A with inhibitors. Emicizumab (Hemlibra), a recombinant, humanized, bispecific monoclonal antibody, restores the function of missing activated factor VIII (FVIII) by bridging FIXa and FX to facilitate effective haemostasis in patients with haemophilia A.
A monoclonal antibody is one that is derived from a single clone of B-cells, and which is specific for a single epitope on a particular antigen.
In the normal clotting cascade, factor VIII acts as a bridge between Factor IXa and factor X, enabling activation of factor X → thrombin generation → clot formation.
Emicizumab is a humanized bispecific antibody, meaning it can bind to two different sites: one arm binds to factor IXa and the other arm binds to factor X, thus mimicking the action of factor VIII, which is absent in haemophilia A patients.
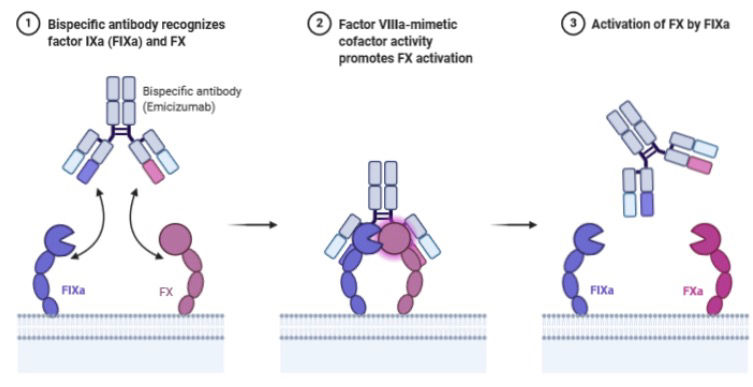
In phase III clinical trials, emicizumab prophylaxis significantly reduced bleeding rates in adolescents and adults with hemophilia A, including those with FVIII inhibitors. Because of the drug's convenient subcutaneous route of administration and relatively infrequent dosing ability (dose every 1, 2, or 4 weeks), emicizumab provides an effective alternative to conventional FVIII replacement products, which tend to be more frequent. The drug has a longer elimination half-life compared to other current treatments and, on average, stays in the body for approximately 4 to 5 weeks.

Emicizumab is a game-changer for patients who have developed inhibitors, since it restores clot formation without the need of fVIII infusions. Since the developed inhibitors act against factor VIII and not a monoclonal antibody, they have no effect on Emicizumab, making it an excellent treatment option for patients with inhibitors.
-By Riya Pandit (LY)


Comments