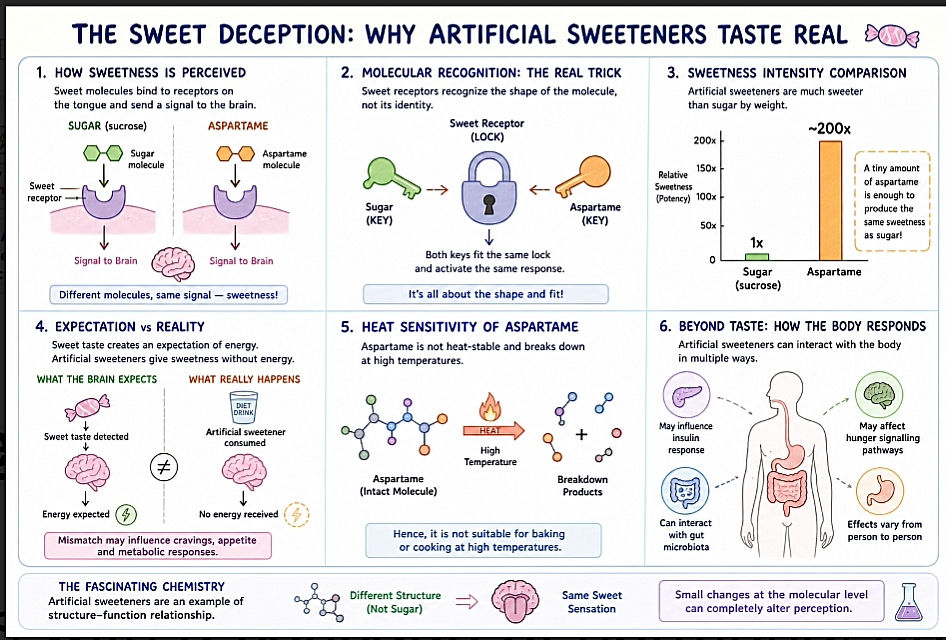
The Chemistry That Made Injections Optional: Understanding SNAC-Mediated Drug Absorption
- ACS BCP
- Apr 5
- 3 min read
The oral administration of peptide-based therapeutics has historically been constrained by extensive enzymatic degradation within the gastrointestinal tract and inherently low epithelial permeability. A major advancement in overcoming these barriers has been the development of sodium N-[8-(2-hydroxybenzoyl) amino] caprylate (SNAC), a carrier-based permeation enhancer introduced by Emisphere in the 1990s. Structurally, SNAC is an N-acetylated amino acid derivative of salicylic acid exhibiting amphiphilic properties, allowing it to interact with both aqueous and lipid environments. Unlike conventional absorption enhancers, SNAC facilitates transcellular drug transport without requiring enteric coating, thereby enabling gastric absorption.
The clinical relevance of SNAC has been most prominently demonstrated in the oral formulation of Semaglutide (marketed as Rybelsus), a glucagon-like peptide-1 (GLP-1) receptor agonist traditionally limited to parenteral administration. Its successful co-formulation represents a significant milestone in the advancement of oral peptide delivery systems.
Mechanism of Action
The absorption-enhancing effect of SNAC is localized, transient, and dependent on co-formulation with the active pharmaceutical ingredient. Upon tablet disintegration in the gastric environment, SNAC facilitates drug stability and absorption through the following coordinated mechanisms:
1.Localized pH Modulation
SNAC induces a localized increase in gastric pH via a buffering effect, thereby reducing the conversion of pepsinogen to pepsin. This minimizes proteolytic degradation and preserves the structural integrity of the peptide during the absorption phase.
2.Promotion of Monomeric Drug Form
SNAC alters the polarity of the surrounding microenvironment, weakening hydrophobic interactions responsible for peptide oligomerization. This promotes the monomeric form of the drug, which exhibits significantly greater membrane permeability.
3.Membrane Fluidization and Transcellular Transport
SNAC interacts with the lipid bilayer of gastric epithelial cells, resulting in a transient increase in membrane fluidity. This reversible structural modification facilitates transcellular passage of the peptide across the gastric epithelium.
Localized and Reversible Action
4.The effects of SNAC are confined to the site of tablet dissolution and are transient in nature, ensuring that epithelial integrity is not permanently compromised.

A critical determinant of SNAC’s functionality is that its absorption-enhancing action is highly dependent on the specific drug with which it is co-formulated, rather than simply co-administered. Effective delivery requires intimate incorporation of SNAC and the active molecule within the same dosage form to ensure synchronized release and localized interaction at the gastric mucosa. Co-administration, in contrast, fails to provide the necessary spatial proximity and temporal coordination required for optimal absorption. This specificity is further illustrated by comparative studies among GLP-1 receptor agonists. While semaglutide demonstrates efficient absorption when co-formulated with SNAC, Liraglutide exhibits less favorable characteristics, including stronger membrane-binding affinity and a higher tendency to oligomerize, which counteracts the monomer-promoting effects of SNAC and limits transcellular transport. Consequently, significantly higher plasma exposure has been observed for semaglutide compared to liraglutide under similar conditions, underscoring the necessity for precise molecular compatibility and tailored co-formulation in SNAC-based delivery systems.
Site of Absorption and Pharmacokinetic Profile
In contrast to most orally administered drugs, which are absorbed in the small intestine, semaglutide co-formulated with SNAC is primarily absorbed in the stomach. Experimental evidence supports this unique absorption pathway, demonstrating localized tablet erosion and enhanced drug concentrations in gastric circulation.
Despite this targeted delivery strategy, oral bioavailability remains low, approximately 0.8–1.4%, and is influenced by dosing conditions such as fasting state and water intake. SNAC itself is rapidly absorbed and eliminated within a few hours, whereas semaglutide exhibits a prolonged half-life of approximately one week, allowing sustained systemic exposure with once-daily dosing.
Conclusion
SNAC represents a pivotal innovation in formulation science, enabling the oral administration of peptide therapeutics through a combination of localized pH modulation, stabilization of monomeric drug forms, and transient enhancement of epithelial permeability. Its successful application in semaglutide underscores the importance of drug-specific compatibility and co-formulation strategies in overcoming fundamental barriers to oral peptide delivery.
References
By Martina Mathews (T.Y Bpharm)


Comments